Introduction
The Ministry of Chemicals and Fertilisers has officially withdrawn the Acid Oil Quality Control Order, 2022, through S.O. 4781(E) dated 22 October 2025.
The decision, issued under the Bureau of Indian Standards (BIS) Act, 2016, means that Acid Oil is no longer under a mandatory BIS certification requirement.
While this move removes an immediate compliance obligation, it also opens a new space for industry, one that values voluntary adherence to standards as a marker of trust, consistency, and market readiness. The BIS certification process itself continues: applications can still be submitted, factory inspections remain active, and testing will follow standard BIS procedures for any applicant who chooses to stay certified under IS 12029:1986 (Acid Oil – Specification).
How the QCO Came Into Play
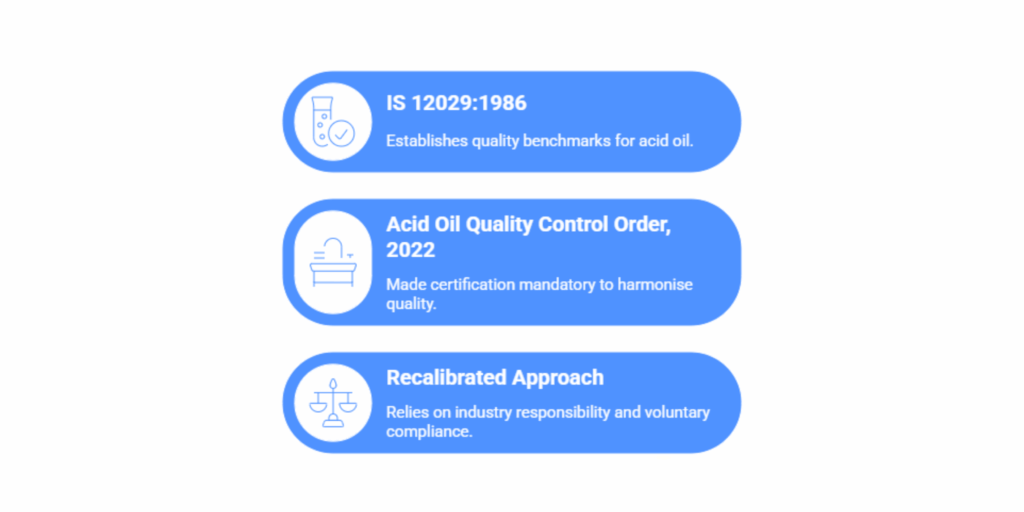
Acid Oil, derived as a by-product from vegetable oil refining, is a critical input in soap manufacturing, animal feed, and other chemical sectors. Because the composition and purity of Acid Oil vary across sources, BIS introduced IS 12029:1986, Acid Oil Specification, to establish a benchmark for key quality parameters, including acid value, moisture, and impurities.
In April 2022, the government issued the Acid Oil Quality Control Order, 2022 (S.O. 1962 (E)), making certification mandatory under this standard. The goal was to harmonise quality within the oleochemical industry, ensuring products met consistent standards during production and trade.
Three years later, after a comprehensive review and consultation with BIS, the Ministry decided that mandatory enforcement was no longer essential in the public interest. The result is a recalibrated approach, one that relies more on industry responsibility and voluntary compliance than on regulation alone.
What the Withdrawal Means in Simple Terms
The withdrawal of the QCO changes how compliance happens, not why it matters.
| Aspect | 2022 QCO | 2025 Update |
|---|---|---|
| Gazette Reference | Ministry of Chemicals & Fertilisers | S.O. 4781(E), 22 October 2025 |
| Standard | IS 12029:1986 – Acid Oil Specification | Same standard (voluntary use) |
| Requirement | Mandatory BIS licence before sale/import | Order rescinded with immediate effect |
| Certification Status | Compulsory | Optional but active |
| Authority | Ministry of Chemicals & Fertilizers | Same Ministry (Dept. of Chemicals & Petrochemicals) |
In essence, the Acid Oil Quality Control Order is no longer enforceable, but BIS certification remains available for companies that wish to maintain or obtain a licence.
How Manufacturers, Importers, and Exporters Can Respond
For Manufacturers
For manufacturers affected by the Acid Oil Quality Control Order, the withdrawal means flexibility without discontinuity. You are no longer legally bound to hold a BIS licence, but you can still choose to pursue or retain one under IS 12029:1986 Acid Oil. Factory inspections, testing, and document evaluations continue as usual for voluntary applicants. This allows manufacturers to demonstrate quality assurance to clients while maintaining internal discipline through BIS-aligned systems.
For Importers
Imports of Acid Oil can proceed without mandatory BIS certification. However, many importers continue to prefer sourcing from BIS-licensed manufacturers to strengthen buyer confidence and streamline trade with regulated industries. Maintaining certificates of analysis and supplier quality documentation remains a practical step for transparency at customs or during audits.
For Exporters
Exporters may view this as an opportunity to stay differentiated. A voluntary BIS mark still communicates reliability and alignment with Indian standards, often valued in tender participation or cross-border trade negotiations.
Why Voluntary BIS Certification Still Matters
Even without a legal requirement, quality assurance never loses value. Voluntary BIS certification under IS 12029:1986 offers several benefits:
- Demonstrates process integrity to partners and regulators.
- Builds readiness for future QCO reissuance.
- Supports reputation and documentation during supply-chain audits.
In short, voluntary compliance becomes a strategic choice, not an administrative burden.
Staying Prepared for What Comes Next
Regulatory cycles evolve; standards withdrawn today may return in updated form later.
Businesses that stay aligned with BIS principles remain more adaptable and credible in the long run.
Here’s how companies can stay prepared:
- Keep internal testing aligned with IS 12029:1986 benchmarks.
- Continue documentation and traceability for every batch.
- Monitor new BIS notifications through verified government sources.
- Engage early if certification becomes relevant again for your product category.
The aim is not to do more paperwork, but to stay ready without disruption.
Turning Change Into Readiness
Every regulatory shift brings both relief and responsibility. With the withdrawal of the Acid Oil Quality Control Order, the emphasis shifts from enforcement to voluntary alignment. For forward-thinking businesses, this is a chance to strengthen quality frameworks and anticipate what comes next, not wait for it.
At NKG Advisory, we help companies interpret such updates clearly and practically, from understanding Gazette notifications to maintaining voluntary BIS certifications. Our approach ensures your quality systems stay audit-ready and adaptable, even as policies evolve.
In compliance, staying informed is the first step to staying prepared. For BIS registration or factory audit support, reach us at www.nkgabc.com or email navraj@nkgabc.com.
To stay updated with more insights on compliance, certifications, and industry trends, explore our blog page or connect with us on LinkedIn for regular updates.